AOAC 2018 Nicotinamide Ribose Analysis by LCMSMS
resources - Recent Conference Materials

A Sensitive, Fast and High-Throughput Quantitative Assay of Nicotinamide Riboside in Microencapsulated Product by LC/MS/MS
Purpose
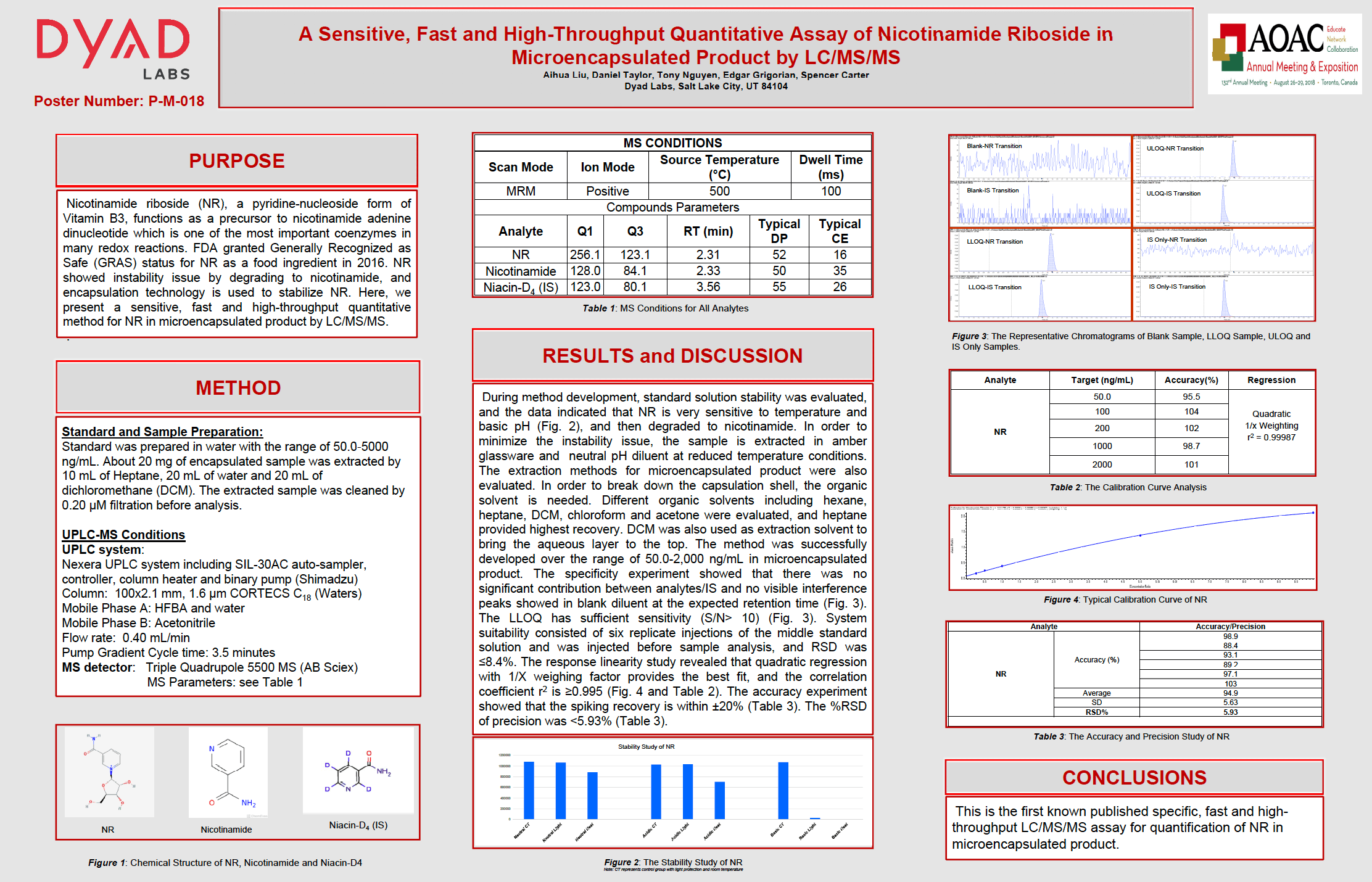
Nicotinamide riboside (NR), a pyridine-nucleoside form of Vitamin B3 functions as a precursor to nicotinamide adenine dinucleotide which is one of the most important coenzymes in many redox reactions. FDA granted Generally Recognized as Safe (GRAS) status for NR as a food ingredient in 2016. NR showed instability issue by degrading to nicotinamide, and encapsulation technology is used to stabilize NR. Here, we present a sensitive, fast and high-throughput quantitative method for NR in microencapsulated product by LC/MS/MS.
Method
Standard and Sample Preparation:
Standard was prepared in water with the range of 50.0-5000 ng/mL About 20 mg of encapsulated sample was extracted by 10 mL of Heptane, 20 mL of water and 20 mL of dichloromethane (DCM). The extracted sample was cleaned by 0.20 μM filtration before analysis.
UPLC-MS Conditions:
UPLC system: Nexera UPLC system including SIL-30AC auto-sampler, controller, column heater and binary pump (Shimadzu)
Column: 100×2.1 mm, 1.6 μm CORTECS C18 ( Waters)
Mobile Phase A: HFBA and water
Mobile Phase B: Acetonitrile
Flow rate: 0 40 mL/min
Pump Cycle time: 3.5 minutes
MS detector: Triple Quadrupole 5500 MS ( AB Sciex)
Results and Discussion
During method development, standard solution stability was evaluated and the data indicated that NR is veryy sensitive to temperature and basic pH (Fig. 2), and then degraded to nicotinamide. In order to minimize the instability issue the sample is extracted in amber glassware and neutral pH diluent at reduced temperature conditions. The extraction methods for microencapsulated product were also evaluated. In order to break down the capsulation shell, the organic solvent is needed. Different organic solvents including hexane, heptane, DCM, chloroform and acetone were evaluated, and heptane provided highest recovery. DCM was also used as extraction solvent to bring the aqueous layer to the top. The method was successfully developed over the range of 50.0-2,000 ng/mL in microencapsulated product. The specificity experiment showed that there was no significant contribution between analytes/IS and no visible interference peaks showed in blank diluent at the expected retention time (Fig. 3). The LLOQ has sufficient sensitivity (S/N> 10) (Fig. 3). System suitability consisted of six replicate injections of the middle standard solution and was injected before sample analysis and RSD was ≤8.4%. The response revealed that quadratic regression with 1/X weighing factor provides the best and the correlation coefficient r2 is 0.995 (Fig.4 and Table 2). The accuracy experiment showed that the spiking recovery is within ±20% (Table 3). The %RSD SD 5.63 RSD% of precision was <5.93% (Table 3).
Conclusion
This first known published specific, fast and high-throughput LC/MS/MS assay for quantification of NR in microencapsulated product.